We pair you with a navigator who understands brain tumor clinical trials and will walk with you from the first search through enrollment.
Thinking About a Clinical Trial? Start Here
Clinical trials test new approaches for treating brain tumors. They follow strict rules to keep participants safe and to see whether a new therapy works. Your navigator can explain the basics in plain language and help you decide if pursuing a clinical trial makes sense for you.
Many brain tumor treatments used today were first studied in clinical trials. Trials can offer access to new therapies that are not available elsewhere. We can help you understand the goals of each study so you can weigh whether it fits your needs and goals.
Each trial has specific criteria about diagnosis, biomarkers, prior treatments, symptoms, and more. Your navigator reviews the details and flags the trials that may fit you. You get clear guidance instead of trying to sort through complex medical language on your own.
If you’re ready to take the next step, we help you prepare. Your navigator can gather and send your records and imaging, walk you through questions to ask, and explain what the study team may need from you. We stay with you as you move through each part of the process.
Clinical trials are always a shared decision between you and your healthcare team. BTN gives you a clear, short list of options to bring to your doctor and helps you talk through the pros and cons. Your doctor leads your care, and we support you with the information that helps you talk together.
Thinking About a Clinical Trial? Start Here

Why Your Decisions Matter
Treatment Choices Today Can Affect Trial Options Tomorrow
Some brain tumor treatments, including medicines like Avastin and devices such as Optune (tumor treating fields), may affect eligibility for future clinical trials. Understanding these tradeoffs can help ensure today’s choices align with your goals. Talk with your navigator and care team about options that support both your needs now and the future.

Connect With a Navigator
Fill out our Get Connected form or call our office at (904) 395-5220. Your dedicated navigator will connect with you to hear your story, review your clinical history and molecular profiling, and talk about whether you are interested in clinical trials.

Complete a consent form to start your trial search
Your navigator will walk you through a quick consent form that allows BTN to gather your medical records and imaging. This gives us the full picture we need to find trials that fit you.

Your navigator searches nationwide for trials that fit you
Your nurse navigator reviews your tumor type, biomarkers, prior treatments, goals, and daily needs. We run a nationwide search, focus on locations that are realistic for you, and answer questions about what life on a clinical trial may look like.
You receive a short, personalized list of trials to bring to your doctor
After your search is complete, your navigator will provide you with a short list of trials that have been vetted and checked for eligibility. You can review them with your navigator then talk with your doctor about which ones fit your goals and what you want moving forward.

We can help you take the next steps
If you decide to pursue a trial, BTN can help with the follow through. We introduce you to the trial’s principal investigator and coordination team, send your medical records for review, and support you as you move between regular care and clinical trial enrollment.
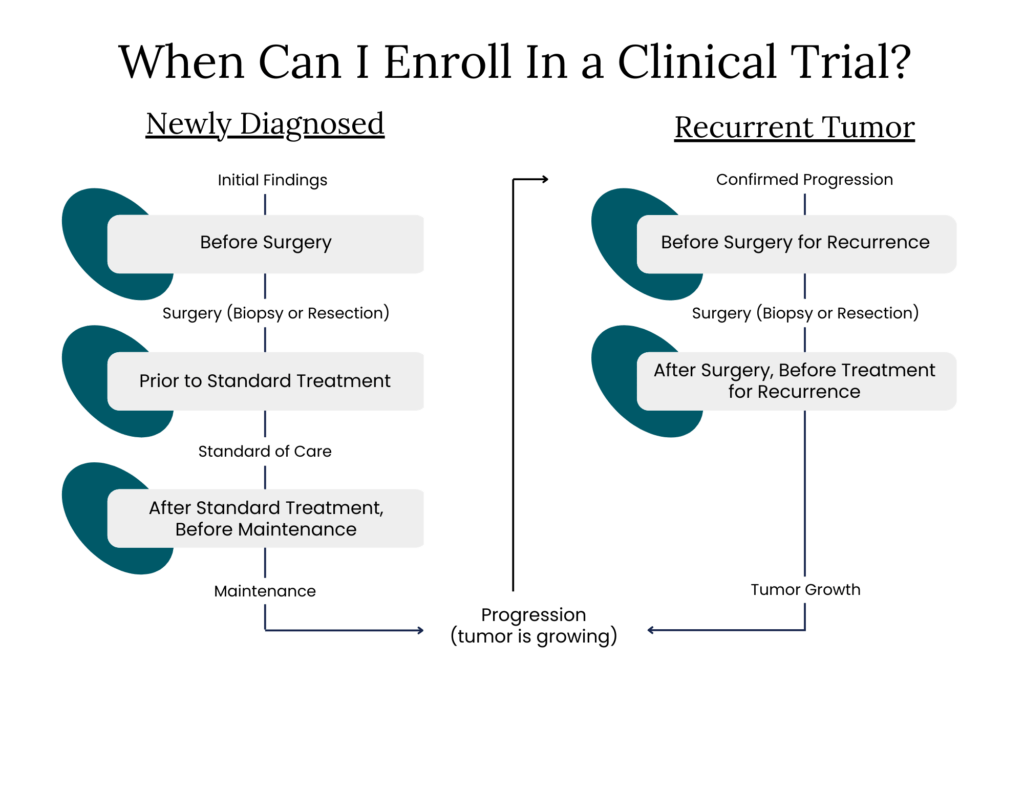
When Can You Enroll in a Clinical Trial?
Some trials open before your first surgery or biopsy, and others open after surgery but before chemotherapy or radiation. These early steps can shape what will be possible later, so it helps to ask about trials as soon as you are diagnosed. Your BTN navigator can walk you through these early timepoints in plain language.
Once surgery or a biopsy is done, there may be a short window before radiation and chemotherapy begin. Some studies use this time to test medicines designed to help prevent fast-growing tumors from spreading or returning. Asking about trials here helps you understand all your choices before treatment starts.
When you finish the standard course of radiation and chemotherapy, there may be trials focused on keeping the tumor from returning. This period is a chance to explore what is available and whether any studies match your tumor type or biomarkers. Your navigator can help you check eligibility and review the details.
A recurrence creates a new set of trial options. This is also an important time to ask about molecular testing, since certain trials require specific biomarkers. BTN can help you understand which studies may fit your personal situation.
When Can You Enroll in a Clinical Trial?
Why Biomarkers Matter
Biomarker Testing Can Open More Doors
Biomarker testing, also called molecular profiling, looks at the genetic details of your brain tumor that cannot be seen on a scan. Knowing these markers can guide treatment choices and may open the door to more options, including clinical trials that are designed for tumors like yours. If you have not had biomarker testing, BTN can help you talk with your doctor about it and use the results in your clinical trial search.